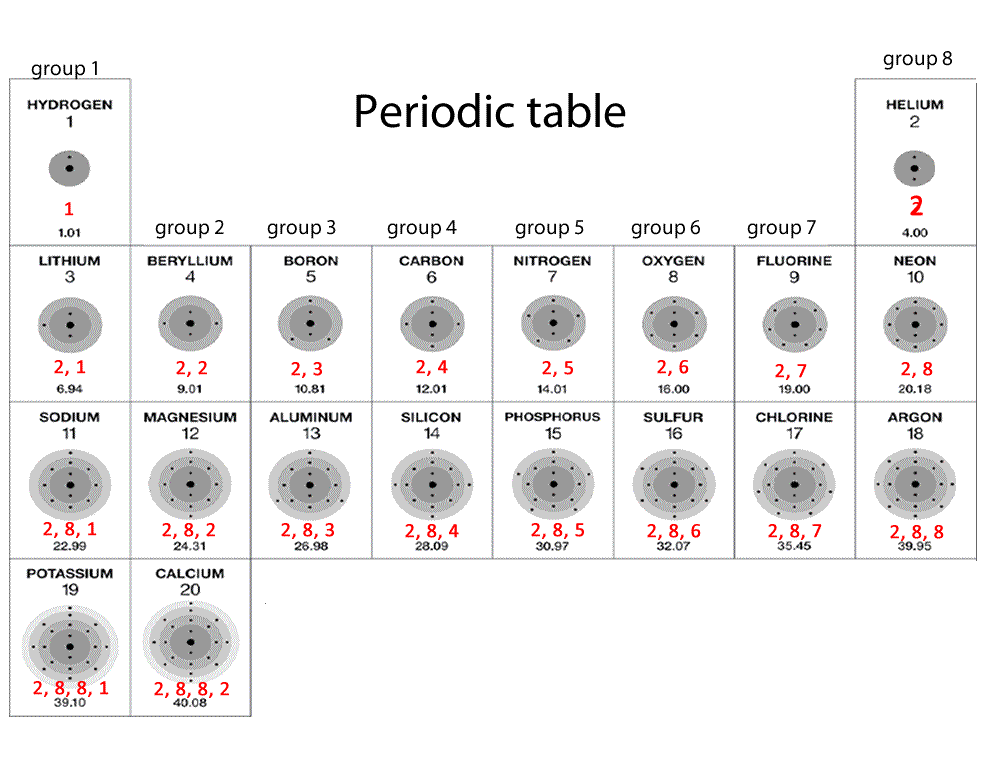
The Pauli Exclusion Principle arises from more than just the electrostatic repulsion of negative electrons: it comes from fundamental physical principles that constrain all subatomic particles. Learn the definition of diatomic elements, where to find diatomic elements on the periodic table, how diatomic elements are formed, and how to remember them. It is an icon of chemistry and is widely used in physics and other sciences. All elements in the alkali metal group occur in nature. Alkali metals are very reactive chemical species that readily lose their one valence electron to form ionic compounds with nonmetals. Alkali metals are any of the elements found in Group IA of the periodic table (the first column). The reason that electrons tend to stay in their separate orbitals rather than piling on top of one another is the Pauli Exclusion Principle, a theorem from quantum mechanics that dictates that no two electrons can ever be in the same place. The periodic table, also known as the periodic table of the elements, arranges the chemical elements into rows ('periods') and columns ('groups'). Chemistry Glossary Definition of Alkali Metal. However, it is possible to remove electrons and not change the identity of an element. Much of the study of chemistry, however, involves looking at what happens when atoms combine with other atoms to form compounds. So the number of electrons is the same as the atomic number. Atoms are the smallest units of matter that still retain the fundamental chemical properties of an element. The halogens (/ h l d n, h e-,-l o-,- d n /) are a group in the periodic table consisting of six chemically related elements: fluorine (F), chlorine (Cl), bromine (Br), iodine (I), and the radioactive elements astatine (At) and tennessine (Ts), though some authors would exclude tennessine as its chemistry is unknown and is theoretically expected to be more like that. 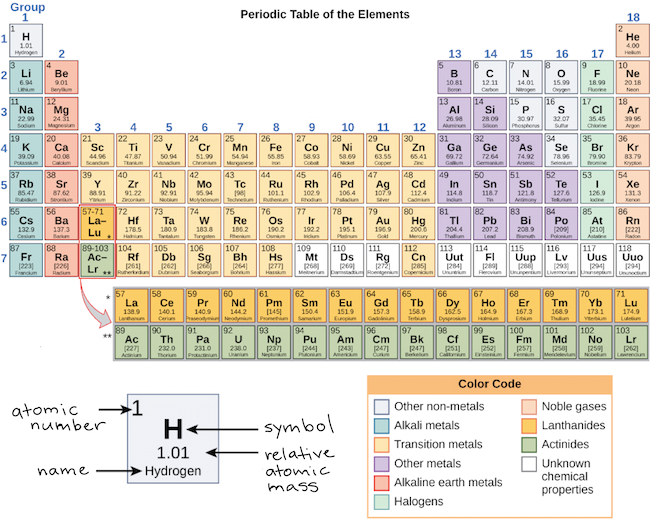
This results in beautiful geometric structures called orbitals that represent the distinct regions around the nucleus that each electron traces out. Molecules are the simplest unit of a covalent compound, and molecules can be represented in many different ways. Together, all of the electrons of an atom create a negative charge that balances the positive charge of. While these electrons all stick within the atom because of their attraction to the protons, they also mutually repel each other, causing them to spread out around the nucleus in regular patterns. Electrons are the negatively charged particles of atom. 
As we know, the positively-charged protons in the nucleus of an atom tend to attract negatively-charged electrons. This first video is of bromine reacting with aluminum.Electron configurations are a simple way of writing down the locations of all of the electrons in an atom. Watch the following two video experiments of \(p\) block elements: A valence electron is an outer shell electron that can participate in a chemical bond with another atom. \) (Credit: Courtesy of NASA Source: (opens in new window) License: Public Domain) Even though protons define the identity of an element, the number of electrons determines the reactivity of each element, allowing the periodic table to sort.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
